Jackson School to Lead $58 Million Effort to Study Potential New Energy Source
October 22, 2014
AUSTIN, Texas — A research team led by The University of Texas at Austin has been awarded approximately $58 million to analyze deposits of frozen methane under the Gulf of Mexico that hold enormous potential to increase the world’s energy supply.

The grant, one of the largest ever awarded to the university, will allow researchers to advance scientific understanding of methane hydrate, a substance found in abundance beneath the ocean floor and under Arctic permafrost.
The Department of Energy is providing $41,270,609, with the remainder funded by industry and the research partners.
In addition to UT Austin’s Institute for Geophysics (UTIG) at the Jackson School of Geosciences, the study includes researchers from The Ohio State University, Columbia University’s Lamont-Doherty Earth Observatory, the Consortium for Ocean Leadership and the U.S. Geological Survey.
“The Department of Energy looks forward to partnering with The University of Texas at Austin and the rest of the project team to plan and execute an outstanding scientific drilling expedition,” said Ray Boswell, program manager at the department’s National Energy Technology Laboratory.
Often referred to as “fire and ice” because of its ability to produce a dazzling flame when lit, methane hydrate is an ice-like solid compound that forms in low-temperature and high-pressure environments where molecules of methane, a chief constituent of natural gas, are trapped within a lattice structure of water molecules.
Estimates vary on the amount of energy that could be produced from methane hydrate worldwide, but the potential is huge. In the Gulf of Mexico, where the team will be sampling, there is estimated to be about 7,000 trillion cubic feet (TCF) of methane in sand-dominated reservoirs near the seafloor. That is more than 250 times the amount of natural gas used in the United States in 2013. Hydrates have the potential to contribute to long-term energy security within the United States and abroad. Many large global economies that lack clean and secure energy supplies have potentially enormous hydrate resources.
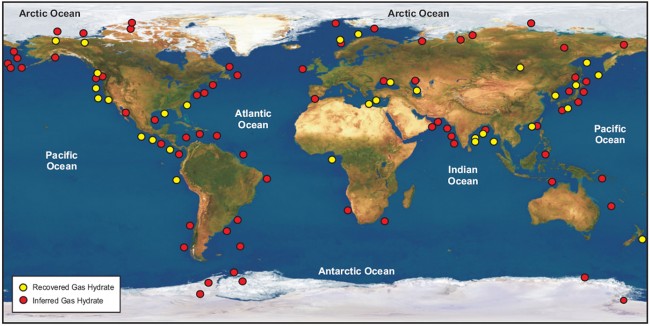
Methane hydrate is stable under high pressure and low temperatures but separates into gas and water quickly when warmed or depressurized, causing the methane to bubble away. This poses technical and scientific challenges to those working to eventually produce energy from the deep-water deposits.
“The heart of this project is to acquire intact samples so that we can better understand how to produce these deposits,” said Peter Flemings, a professor and UTIG research scientist and the project’s principal investigator.
The four-year project will be the first in the offshore United States to take core samples of methane hydrate from sandstone reservoirs, Flemings said, a delicate task that requires transporting samples from great depths to the surface without depressurizing them.

Carlos Santamarina, a professor at the Georgia Institute of Technology and a leading methane hydrate expert, said pressure core sampling is vital to gaining a better scientific understanding of hydrate-bearing sediments.
“The technique is like taking a specimen inside a pressure cooker from thousands of feet below sea level, and bringing it to the surface without ever depressurizing the pressure cooker,” said Santamarina. “With this technology, the sediment preserves its structure and allows us to determine all the engineering properties needed for design.”
It is not currently economically or technically feasible to produce substantial amounts of energy from methane hydrate, but Flemings said that could change as the science improves and world energy demand increases.
“This could be analogous to gas or shale oil 20 or 30 years ago,” he said. “None of us thought we were going to produce any hydrocarbons out of shales then.”
Santamarina said this project is critical for the United States to maintain world leadership in methane hydrate research. Other countries with high energy demands or limited resources — Japan, South Korea, India and China — also have active research programs.
In addition to its enormous potential as an energy resource, methane hydrates may play a role in past and future climate change, and better understanding the marine deposits will further scientific understanding of these processes.
“I think methane hydrates are one of the most fascinating materials on the planet,” Flemings said. “They store energy, they look like ice but burn, they may impact climate, and they may play a role in submarine landslides.”
Santamarina said he believes the right team has been assembled to tackle the complex challenges.
“The best people in the world will be involved in this project,” he said. “It is exceptional that UT is going to lead this effort.”
For more information, contact: Anton Caputo, Jackson School of Geosciences, 512-232-9623.